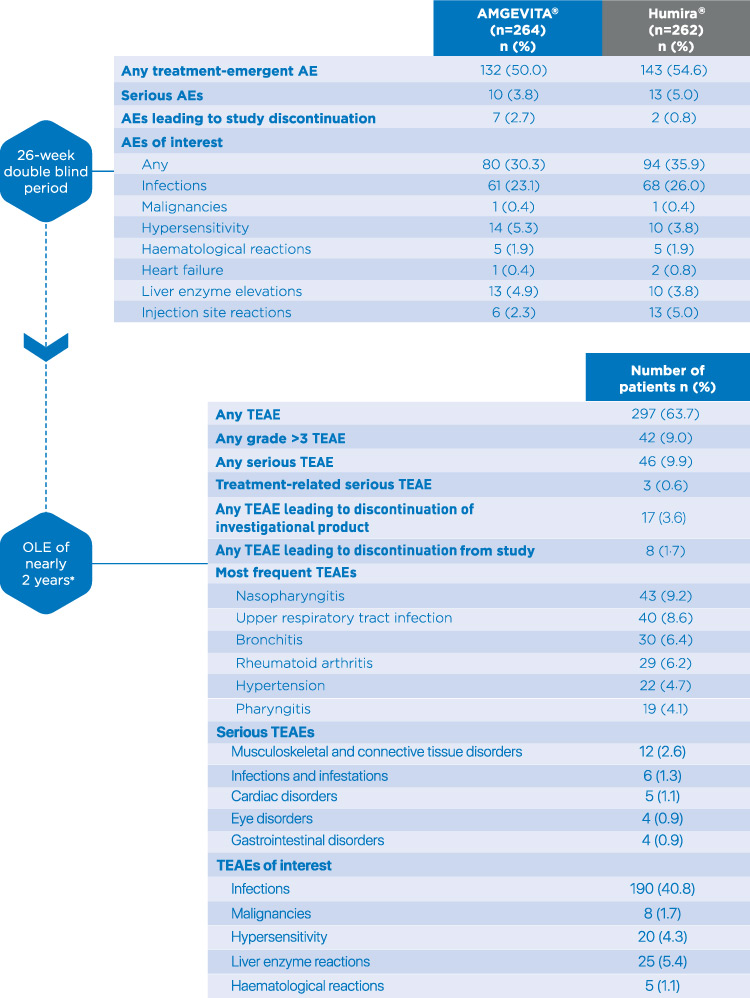
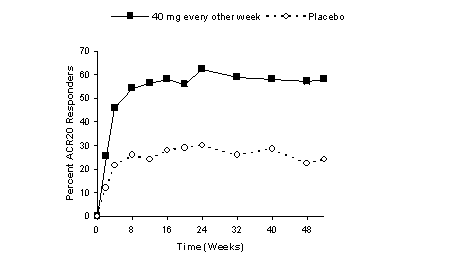
Study design. *Initial dose of adalimumab was 80 mg, followed by 40 mg... | Download Scientific Diagram

CVS Health and Sandoz launch private label biosimilar products | Eddy Palacios posted on the topic | LinkedIn
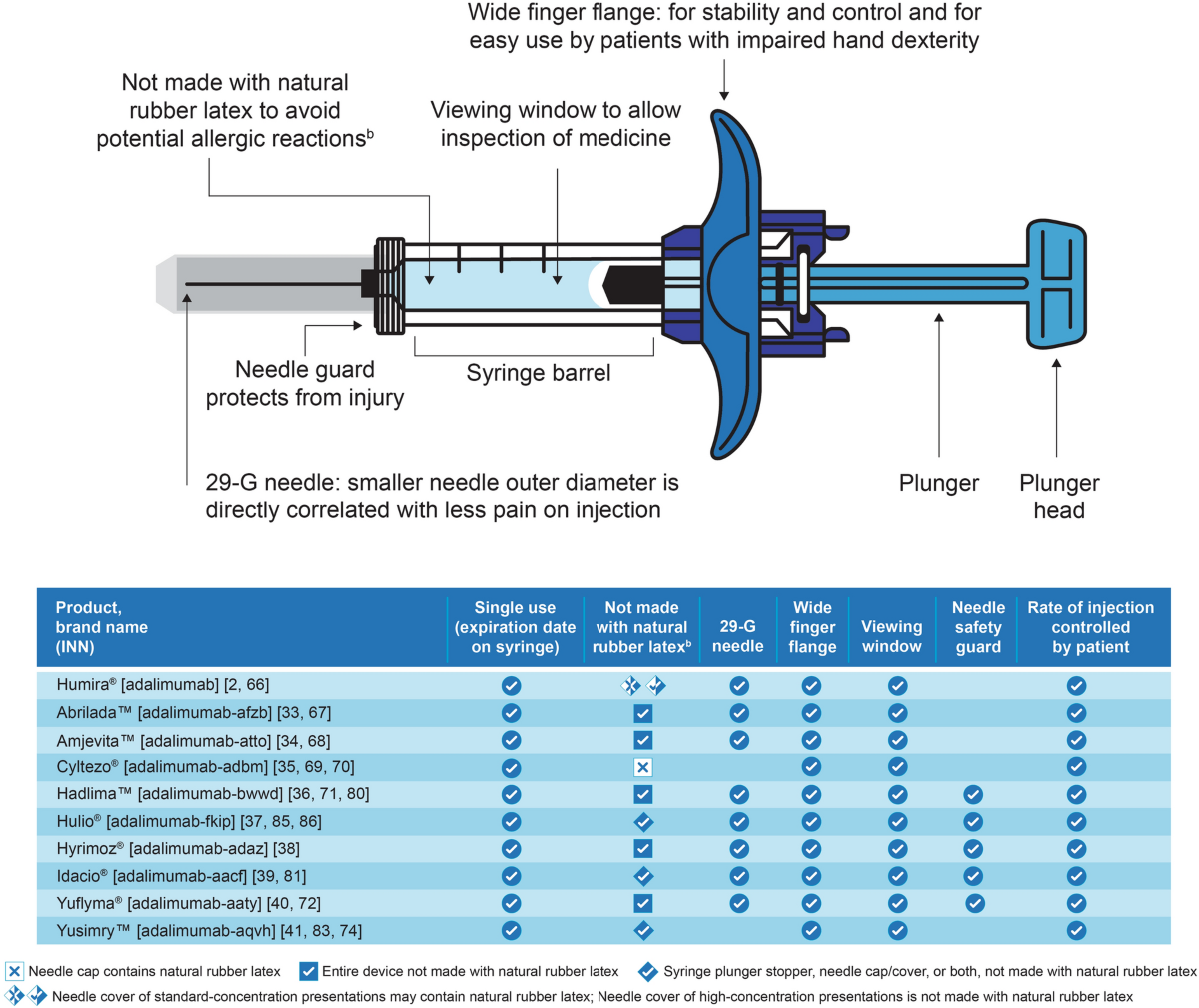
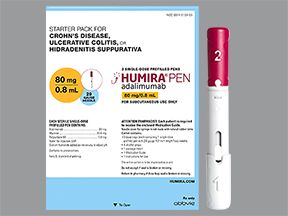
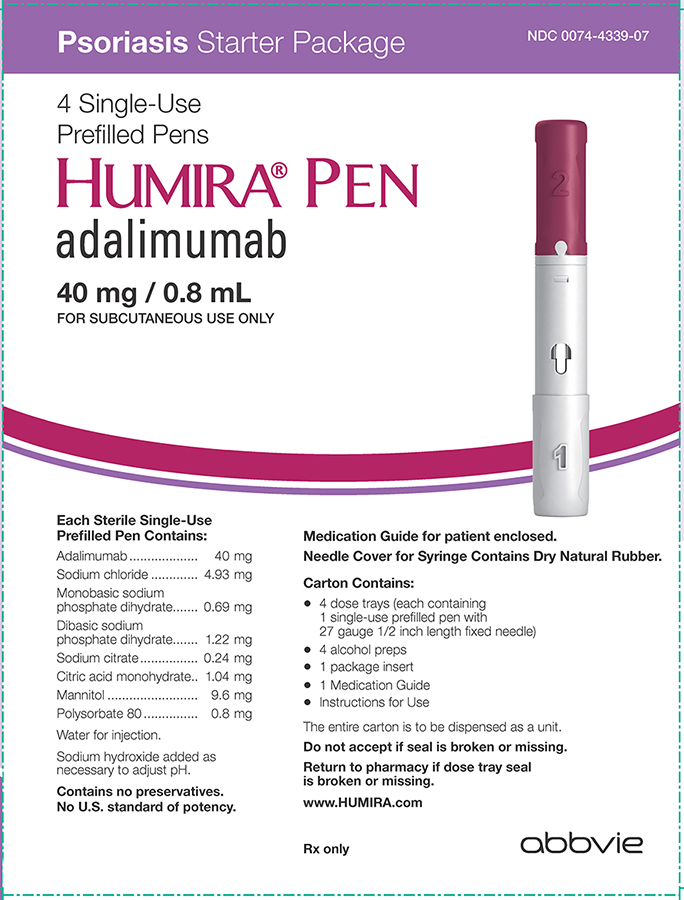
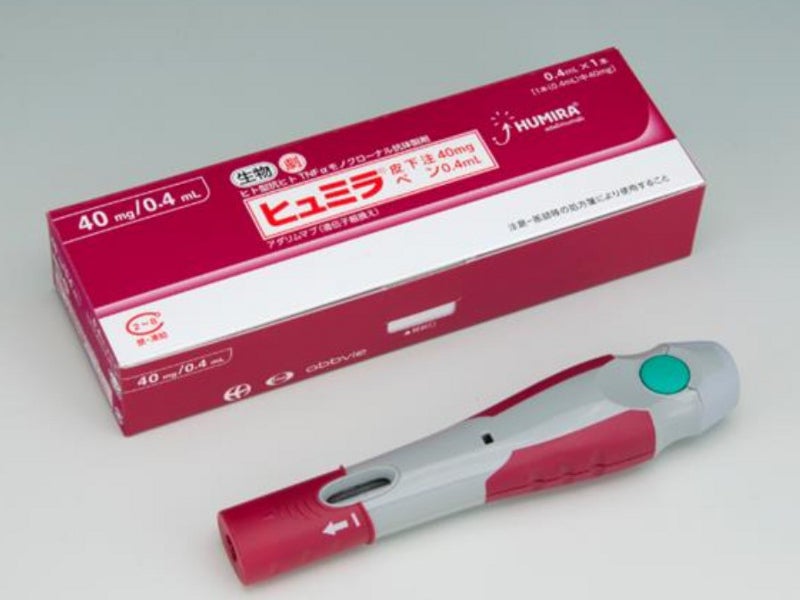
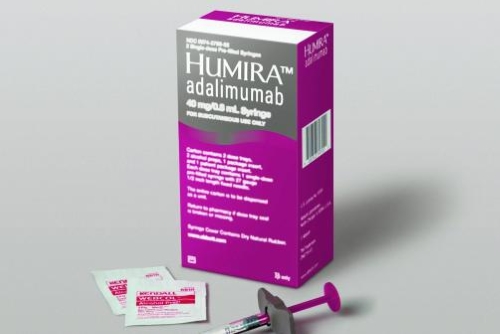
Relevance of Adalimumab Product Attributes to Patient Experience in the Biosimilar Era: A Narrative Review | Advances in Therapy